What is meant by P-type material?
Solution : Doping is done in semiconductors to increase the number of mobile electrons //holes and therefore to increase the conductivity of the semiconductors.
Which is better p-type or n-type semiconductor?
Impurities based on the number of valence electrons can be n-type (5) or p-type (3). The n-type tend to be a better choice due to reduced LID (Light Induced Degradation) & increasing durability and performance compared to p-type.
Which has more p-type or n-type holes? The added impurity in the p-type semiconductor provides extra holes known as an Acceptor Atom, while the n-type semiconductor impurity provides extra electrons and is called a Donor atom.
Why does n-type semiconductor has more conductivity than p-type?
Why is the conductivity of n-type semiconductors greater than that of p-type semiconductors even when both have the same level of doping? Solution : The conductivity of n-type semiconductors is greater because the mobility of electrons is more than that of holes.
Why do we prefer n-type semiconductors?
Because in N semiconductors the majority carries are electrons and holes in ptype semiconductors, electrons have more mobility than holes and therefore we have more current in Ntype than ptype for the same dimensions and the same voltage.
Which type of semiconductor is preferred?
Silicon is the most widely used type of semiconductor material. Its main advantage is that it is easy to fabricate and provides good overall electrical and mechanical properties.
Why is n-type semiconductor better?
This impurity leads to better conductivity among semiconductors. Impurities based on the number of valence electrons can be n-type (5) or p-type (3). The n-type trend is a better choice due to the reduction of LID (Light Induced Degradation) and the increase in durability and performance compared to the p-type.
Which is more conductivity n-type or p-type semiconductor?
The conductivity of n-type semiconductors is greater than that of p-type semiconductors because the mobility of electrons is greater than that of holes.
Which has more conductivity p-type or n-type?
Since free electrons have more mobility than holes, therefore, an n-type semiconductor has a higher conductivity than that of a p-type semiconductor.
Which semiconductor conductivity is high?
| Material | Resistivity (Ω-cm) | Conductivity (Ω-1-cm-1) |
|---|---|---|
| Copper (tempered) | 1.72 x 10-8 | 5.95 × 107 |
| Aluminum | 2.65 x 10-8 | 3.77 × 107 |
What is impurities in semiconductor?
Two types of impurities are added to semiconductors. They are pentavalent and trivalent impurities. Pentavalent impurities. Pentavalent impurity atoms have 5 valence electrons. The various examples of pentavalent impurity atoms include Phosphorus (P), Arsenic (As), Antimony (Sb), etc.
What impurities are added in semiconductors? The addition of pentavalent impurities such as antimony, arsenic or phosphorus contributes free electrons, greatly increasing the conductivity of the intrinsic semiconductor. Phosphorus can be added by diffusion of phosphine gas (PH3).
What are impurities in physics?
January 2021. In chemistry and materials science, impurities are chemical substances inside a restricted amount of liquid, gas or solid, which are different from the chemical composition of the material or compound.
What is impurity in n-type semiconductor?
An n-type semiconductor is an intrinsic semiconductor doped with phosphorus (P), arsenic (As), or antimony (Sb) as an impurity. Group IV silicon has four valence electrons and Group V phosphorus has five valence electrons.
Why impurities are used in semiconductor?
Impurities are added to a semiconductor to actually increase the electrical conductivity. The process of adding impurity to the semiconductor to increase its ability to conduct electricity is known as doping and the impure semiconductor is known as doped semiconductor.
What are impurities in electronics?
Two types of impurities are added to semiconductors. They are pentavalent and trivalent impurities. Pentavalent impurity atoms have 5 valence electrons. The various examples of pentavalent impurity atoms include Phosphorus (P), Arsenic (As), Antimony (Sb), etc.
Why are impurities added?
The addition of small amounts of impurities allows the control of semiconductor conductivity: low donors, such as phosphorus in silicon, produce n-type (electron-carrying) conductivity, and low acceptors, such as -boron in silicon, produce p-type conductivity (carried by holes).
How do you make p-type and n-type?
The formation of p-type semiconductor material can be done by adding group III elements. Similarly, the n-type semiconductor material can be formed by adding group V elements.
What is P-type and n-type? The majority carriers in a p-type semiconductor are holes. In an n-type semiconductor, a pentavalent impurity from the V group is added to the pure semiconductor. Examples of pentavalent impurities are Arsenic, Antimony, Bismuth etc. Pentavalent impurities provide extra electrons and are called donor atoms.
How a N-type is formed?
An n-type semiconductor results from the implantation of dopant atoms that have more electrons in their outer shell (bonding) than silicon. The resulting semiconductor crystal contains extra, or free, electrons that are available to conduct current.
What makes something an N-type semiconductor?
What is an n-type Semiconductor? An n-type semiconductor is an intrinsic semiconductor doped with phosphorus (P), arsenic (As), or antimony (Sb) as an impurity. Group IV silicon has four valence electrons and Group V phosphorus has five valence electrons.
How is an N-type material created?
An N-type semiconductor is a type of material used in electronics. It is made by adding an impurity to a pure semiconductor such as silicon or germanium. The impurities used can be phosphorus, arsenic, antimony, bismuth or some other chemical element. These are called donor impurities.
How do you create a P-type material?
To make a p-type semiconductor extra materials such as boron or aluminum are added to the silicon. These materials have only three electrons in their outer shell. When the extra material replaces some of the silicon it leaves a hole where the fourth electron would be if the semiconductor were pure silicon.
How n-type and p-type semiconductors are prepared?
Doping Semiconductors Pentavalent impurities Impurity atoms with 5 valence electrons produce n-type semiconductors by contributing extra electrons. Trivalent impurities Impurity atoms with 3 valence electrons produce p-type semiconductors by producing a “hole” or electron deficiency.
How p-type and n-type semiconductors are formed?
A p-type semiconductor is created when group III elements are doped for a complete semiconductor material. Conversely, an n-type semiconductor is created when group V elements are doped to an intrinsic semiconductor.
How are p and n type materials created? A PN-junction is formed when n-type and p-type material are joined together to create a semiconductor diode. It all comes down to the p-n junction. N-type silicon has extra electrons and there are atoms on the p side that need electrons, so electrons migrate across the junction.
How N-type semiconductor are formed?
An n-type semiconductor results from the implantation of dopant atoms that have more electrons in their outer shell (bonding) than silicon. The resulting semiconductor crystal contains extra, or free, electrons that are available to conduct current.
What is meant by n-type of semiconductor?
What is an n-type Semiconductor? An n-type semiconductor is an intrinsic semiconductor doped with phosphorus (P), arsenic (As), or antimony (Sb) as an impurity. Group IV silicon has four valence electrons and Group V phosphorus has five valence electrons.
What is meant by n-type?
Definition of type N ÄntÄ«p. Made of a material, usually a semiconductor such as silicon, that has been doped with impurities so that it has an excess of conductive electrons. 3. Made of a material, usually a semiconductor such as silicon, that has been doped with impurities so that it has an excess of conductive electrons.
Sources :


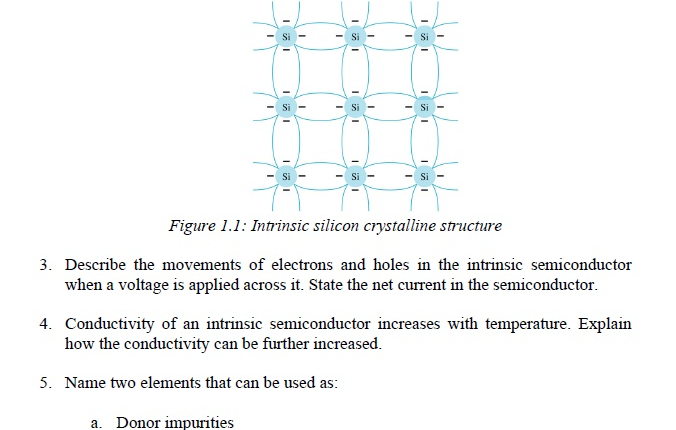

Comments are closed.